Applied BioCode has submitted its Respiratory Pathogen Panel (RPP), which detects viruses and bacteria that cause upper respiratory infections, for 510(k) clearance with the U.S. Food and Drug Administration (FDA). It hopes to launch the product in December.
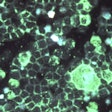
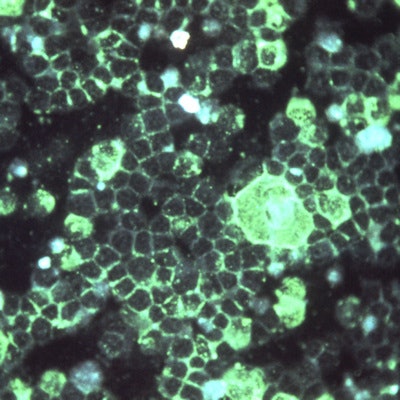
 Indirect immunofluorescence microscopy of RSV. Image courtesy of the U.S. Centers for Disease Control and Prevention/Dr. Craig Lyerla.
Indirect immunofluorescence microscopy of RSV. Image courtesy of the U.S. Centers for Disease Control and Prevention/Dr. Craig Lyerla.The BioCode RPP works with the company's MDx-3000 molecular system. It tests swabs from the back of the nose and throat for 20 common viruses and bacteria, including influenza A, influenza B, respiratory syncytial virus (RSV), and Chlamydia pneumoniae. Up to 188 samples may be processed during an eight-hour shift, according to the company.
The filing for FDA clearance of the respiratory panel was supported by a study of more than 2,600 samples evaluated prospectively at five geographically distributed laboratories in the U.S. This is the second panel developed for the MDx-3000 system. Applied BioCode's Gastrointestinal Pathogen Panel, which tests for 17 common bacteria, viruses, and parasites that cause infectious diarrhea, was cleared by the FDA in 2018.
The company is developing a third panel for detecting agents that cause sexually transmitted infections.