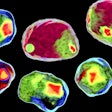
Scientists are obtaining vital data on disease processes via a “revolutionary” new scanning method that shows cellular structures in 3D and at an “unprecedented” level of detail, say researchers.
The system, called wildDISCO, makes use of standard antibodies to map its entire body using fluorescent markers. wildDISCO can create whole-body “atlases” focusing on the nervous system, lymphatic vessels, blood vessels, and immune cells to highlight normal structures and tumors in early stages of formation.
Researchers led by Prof. Ali Ertürk, head of the Institute for Tissue Engineering and Regenerative Medicine at Helmholtz Munich, say their work, reported in Nature Biotechnology, facilitates modeling of complex biological systems and diseases while reducing animal use.
The research team has mapped tumor-associated lymphoid structures (TLS) in relation to breast cancer, which they say will “provide insights into how these structures affect the immune response to tumors.”
“Knowing where each protein is expressed in the body is essential for developing a comprehensive understanding of how the body works and what goes wrong in complex diseases,” Ertürk said.
In the past, scientists relied on genetically modified animals or specialized labels to make specific structures and cells of interest visible in the entire body of an animal. However, these approaches are expensive and time-consuming to create.
In previous work, Prof Ertürk and colleagues developed uDISCO, a clearing method based on organic solvents to render opaque tissues and the whole bodies of dead mice transparent, but it was not possible to image throughout the entire animal using uDISCO.
The wildDISCO system is a whole-body IgG antibody immunolabeling method aided by virtual reality (VR) visualization that overcomes existing technical limitations, say the researchers. It facilitates imaging of peripheral nervous systems, lymphatic vessels and immune cells in whole mice at cellular resolution by labeling diverse endogenous proteins. A fluorescence signal is detected where a specific antibody is bound to the structure, molecule, or cell of interest.
A crucial factor in the project was finding a way to uniformly distribute a fairly large antibody homogenously throughout every cell of an animal. Prof. Ertürk and his team identified a specific compound that enhances cell membrane permeability and facilitates deep and even penetration of standard antibodies without aggregation. They are now looking to further enhance the system’s capabilities and train artificial intelligence (AI) algorithms on the large datasets it produces. Ertürk said that harnessing the power of AI could help to develop new treatments more efficiently “via computational predictions without the need for further animal experiments.”
An atlas of high-resolution wildDISCO images showcasing mouse nervous, lymphatic, and vascular systems. is accessible.
“Our online atlases have already generated data for published papers. Other scientists have obtained critical data saving time, resources, and reducing animal use,” write Drs. Hongcheng Mai and Jie Luo, two postdoctoral researchers at Helmholtz Munich, in Nature Biotechnology.