The analysis of tissue samples has paved the way for a molecular signature for small cell neuroendocrine cancers that could be used to develop treatments that work across tumor types, researchers from the University of California, Los Angeles (UCLA) reported in the July 8 issue of Cancer Cell.
The signature was developed through a computational study of 10,000 patient tumor samples in 35 tumor types -- including small cell neuroendocrine cancers (also called small cell cancers) and non-small cell cancers. Genes that strongly contribute to the small cell neuroendocrine signature include chromogranin A (CHGA), insulinoma-associated protein 1 (INSM1), achaete-scute homolog 1 (ASCL1), neuronal differentiation 1 (NEUROD1), seizure protein 6 (SEZ6), internexin neuronal intermediate filament protein alpha (INA), and NK2 homeobox 2 (NKX2-2), the researchers noted.
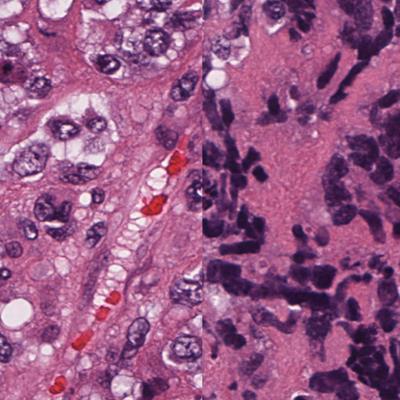
 Microscopic images show a non-small cell breast cancer (left) and small cell neuroendocrine breast cancer (right). Small cell neuroendocrine cancers typically feature smaller cells and larger nuclei in relation to cell size. Image courtesy of Cancer Cell/UCLA Broad Stem Cell Research Center.
Microscopic images show a non-small cell breast cancer (left) and small cell neuroendocrine breast cancer (right). Small cell neuroendocrine cancers typically feature smaller cells and larger nuclei in relation to cell size. Image courtesy of Cancer Cell/UCLA Broad Stem Cell Research Center."It is important to note that any one cancer incidence may contain only a subset of these markers and hence be missed by traditional classification schemes based on only a few markers," wrote Thomas Graeber, PhD, a professor of molecular and medical pharmacology at the UCLA Metabolomics Center, and colleagues (Cancer Cell, July 8, 2019, Vol. 36:1, pp. 17-34.E7).
The signature could be used to develop drug treatments for a range of small cell cancers, which are more aggressive and difficult to treat; small cell types have been reported in lung and prostate cancers, among others.
Interest in treatments that are tissue-agnostic -- that is, they target a particular genetic signature, rather than being based on tissue type or location -- has increased in recent years along with the push for precision medicine and growing knowledge about tumor biology through genomic sequencing studies. The programmed cell death 1 (PD-1) inhibitor pembrolizumab (Keytruda, Merck) became the first drug approved by the U.S. Food and Drug Administration for a tumor-agnostic indication in May 2017, when it was cleared for tumors with high microsatellite instability or DNA mismatch repair deficiency.
Guided by pathologists' observations
The UCLA researchers said their analysis was guided by pathologists, who had reported common features in a variety of different small cell cancers.
"The highly convergent molecular profiles of [small cell neuroendocrine] tumors reported here match well-documented pathology findings of related morphologies and biomarkers across different tissue types," Graeber and colleagues wrote.
It has been unclear how the cancers develop; there are indications that resistance to treatment plays a role.
"This has important consequences in that [small cell neuroendocrine cancers], once considered rare, may become increasingly common with the emergence of resistance cases from targeted therapies," they wrote.
Currently, there are no effective therapies for small cell neuroendocrine cancers, which are associated with early and widespread metastases and, consequently, poor prognosis, the authors noted.
In the study, small cell neuroendocrine features were reported across multiple tumors, including in "purportedly non-[small cell neuroendocrine] primary tumors," which suggests they had been missed in original pathology reports, the group wrote.
Blood cancer connection
In addition to reporting about the development of the molecular signature, the researchers noted that small cell neuroendocrine and blood cancers have common RNA interference susceptibilities and drug sensitivities -- an unexpected finding. This means that drugs used to treat hematologic malignancies, such as inhibitors of histone deacetylase (HDAC) and B-cell lymphoma 2 (BCL-2), have potential in small cell neuroendocrine cancers, perhaps used in combination. BCL-2 inhibitors have been tested in preclinical models and early clinical studies of small cell lung cancer.
The size of the potential patient population with the molecular signature across cancers is unclear, Graeber commented by email to LabPulse.com. The researchers do believe the number is growing, because there is evidence that targeted therapies for lung cancer and prostate cancer can lead to small cell-based resistance mechanisms.
"We anticipate that this may be seen in additional cancer types, and in the context of other targeted therapies," Graeber said.
Building better lab models
The UCLA team is now building better laboratory models of transdifferentiation to a small cell phenotype, so they can explore therapeutic ways to block this therapy escape route, while also following up on the lead that small cell tumors share vulnerabilities with blood cancers, he explained.
"Since therapeutic options for blood cancers have advanced over the years, especially in comparison to options for small cell cancers, we want to explore how this unexpected connection might lead to better small cell-targeted approaches," Graeber said.