Researchers in China have found that reducing the methylation of a key messenger RNA can promote the migration of macrophages into the brain and ameliorate symptoms of Alzheimer’s disease in mice.
The study, published Tuesday in PLOS Biology, illuminates one pathway for the entrance of peripheral immune cells into the brain, and may provide a new target for Alzheimer’s disease treatment.
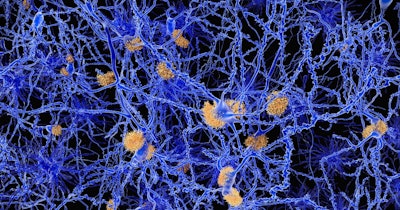
A presumed trigger for the development of Alzheimer’s disease is the accumulation of proteinaceous, extracellular amyloid-beta plaques in the brain. High amyloid-beta levels in mice lead to neurodegeneration and cognitive decline similar to human Alzheimer’s disease. Therefore, amyloid-beta reduction is a major goal in the development of new treatments.
One potential pathway for getting rid of amyloid-beta is the migration of blood-derived myeloid cells, which serve as immune responders, into the brain. These cells mature into macrophages which, along with resident microglia, can consume amyloid-beta. The cells’ migration is a complex phenomenon controlled by multiple interacting factors, but a potentially important factor is the methylation of messenger RNA (mRNA) within the myeloid cells.
The most common type of mRNA methylation, called m6A, is carried out by the enzyme METTL3. The researchers sought to learn whether reduced mRNA methylation leading to METTL3 deficiency in myeloid cells had an effect on cognition in mice with Alzheimer’s disease-like symptoms. They found that mice treated to become METTL3 deficient performed better on various cognitive tests, an effect that could be inhibited when the researchers blocked myeloid cell migration into the brain.
To learn how decreased mRNA methylation promoted myeloid cell migration, the researchers discovered a complex mechanism. Through analysis of mRNA expression patterns and other techniques, they showed that METTL3 depletion reduced the activity of a key m6A reader protein, which recognizes m6A-modified mRNAs and promotes their translation into protein. That led to a decline in another protein, which in turn inhibited the production of yet another protein, called ATAT1. Loss of ATAT1 reduced the attachment of acetyl groups to microtubules; that reduction promoted the migration of the myeloid cells into the brain. The myeloid cells’ maturation into macrophages provided increased clearance of amyloid-beta plaques, and improved cognition in mice.
While much about this complex Alzheimer’s disease pathway remains to be explored, the research indicates that this path does promote the immune-responding myeloid cell migration, and the resultant decrease of toxic plaques. “Our results suggest that m6A modifications are potential targets for the treatment of Alzheimer’s disease,” the researchers concluded. However, they cautioned, “Because mRNA methylation has a fundamental effect on a wide variety of downstream targets, effective drug development within this pathway may require moving further downstream to avoid unwanted effects.”