The U.S. Food and Drug Administration (FDA) has approved Vertex Pharmaceuticals’ and CRISPR Therapeutics’ CRISPR-based gene-editing therapy for the treatment of vaso-occlusive crises (VOCs) in patients with sickle cell disease (SCD). It is the first CRISPR-based gene therapy to receive FDA approval.
Casgevy (exagamglogene autotemcel), also known as exa-cel, was approved for the treatment of SCD and transfusion-dependent β-thalassemia in the U.K. in late November.
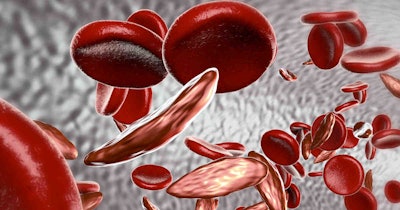
Casgevy is now approved in the U.S. to treat patients 12 years of age and older who have recurrent VOCs from SCD. These recurrent VOCs are often debilitating and may lead to life-threatening disabilities and early death.
An FDA advisory committee meeting was held on October 31 to discuss and make recommendations on Casgevy.
In testing, of the 31 patients participating in the study for which sufficient follow-up time has passed, 29 (93.5%) have remained free of VOC episodes for at least a year. None of the patients have had graft failure or graft rejection.
Additionally, the FDA has approved Bluebird Bio’s Lyfgenia, a cell-based gene therapy that uses a lentiviral vector for genetic modification for the treatment of patients 12 years of age and older with SCD and a history of severe vaso-occlusive events (VOEs).
In the Phase I/II study for Lyfgenia, severe VOEs were eliminated for 30 of 32 (94%) of evaluable participating patients, and 28 of 32 (88%) of the patients have remained completely free of VOEs between 6 and 18 months after treatment.
The applications for both Casgevy and Lyfgenia received Priority Review, Orphan Drug, Fast Track, and Regenerative Medicine Advanced Therapy designations from the FDA.
“Sickle cell disease is a rare, debilitating and life-threatening blood disorder with significant unmet need, and we are excited to advance the field especially for individuals whose lives have been severely disrupted by the disease by approving two cell-based gene therapies today,” said Dr. Nicole Verdun, director of the Office of Therapeutic Products within the FDA’s Center for Biologics Evaluation and Research. “Gene therapy holds the promise of delivering more targeted and effective treatments, especially for individuals with rare diseases where the current treatment options are limited.”