Dear LabPulse Member,
This past week brought some encouraging developments for clinical laboratories coping with COVID-19 in the U.S.
For example, the U.S. Department of Health and Human Services launched the National Testing Implementation Forum, a body that will allow stakeholders a voice on strategies for SARS-CoV-2 surveillance, diagnostics, and supplies.
This is a welcome development for organizations that represent pathology personnel and clinical laboratories, which have been clamoring for more effort and cohesion at the national level to address supply shortages and other critical issues.
A recently published study in the Lancet Public Health journal drove home the importance of rapid testing for the novel coronavirus to enable contact tracing and isolation of those at risk. After the onset of symptoms, testing should be conducted within three days, researchers advised.
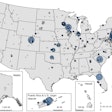
According to the U.S. Centers for Disease Control and Prevention, there were more than 3.9 million cases of COVID-19 and 143,000 total deaths in the country as of July 23. The reopening of workplaces in parts of the U.S. and the risk for people who participated in recent protests are among the factors driving increased demand for testing.
Quest Diagnostics has been speaking out in recent weeks about demand outstripping supply. On July 18, the company's SARS-CoV-2 real-time reverse transcription polymerase chain reaction test became the first to receive an emergency use authorization from the U.S. Food and Drug Administration (FDA) for testing pooled samples. Pooled testing has become a centerpiece in the U.S. strategy for expansion of testing, and in a statement about the clearance the FDA emphasized its willingness to work with diagnostic test developers to introduce new options. The agency has also released new guidance that it believes will help expand the availability of transport media for clinical specimens.
Non-COVID-19 highlights from our recent coverage include reports on how a strategy for mailing fecal immunohistochemical tests to Medicaid patients could boost colorectal cancer screening rates and the deployment of a deep-learning system developed at Google for Gleason grading of prostate cancer biopsy specimens.
Finally, dear member, I have some personal news to share. I am soon moving on from LabPulse.com and this is my last editor's note to you. It's been fascinating for me to see how the image of lab professionals has changed from one of obscurity when this website launched in April 2019 to the highest profile of perhaps all medical specialties during the COVID-19 pandemic.
Since the crisis started, change has been the name of the game. I moved out of the office and started working from home, had a backroom haircut, and took a drive-through nasal swab molecular test. I picked up the ukulele in pandemic-style Zoom lessons and now know a handful of chords, which is enough to accompany myself singing classic country songs as the public emergency continues. It's been a pleasure getting to know the specialty of lab medicine.