Advances in technology, coupled with a regulatory waiver, have given pathologists the option to work remotely, often from the safety of their home-based medical offices, during the COVID-19 crisis.
But telepathology, or remote pathology, can only be efficient if the pathologists at home, or elsewhere away from the lab, can qualitatively make as exact a diagnosis of tumors and other diseases on the computer screen as they can using a lab microscope.
On March 26, the federal government announced a temporary waiver giving pathologists regulatory flexibility in terms of where they work, with temporary relief from CLIA regulations that require pathologists to perform diagnostic tests at certified facilities. The waiver also allows pathologists to perform remote sign-out. The U.S. Centers for Medicare and Medicaid Services (CMS) indicates it is exercising discretion when enforcing regulations to ensure pathologists may review pathology slides remotely using certain guidelines.
Validation and other issues
In addition to concerns about image quality, the process of transitioning from reading slides on a microscope to reading them electronically on a monitor necessitates addressing a number of issues including cybersecurity, system validation, and cost. And, it's important to plan ahead.
"The key to success for digital pathology would be to have a system in place (at a central lab) that's already been validated," Dr. Eric Glassy, medical director at the Affiliated Pathologists Medical Group in Rancho Dominguez, CA, said in an interview.
Verification and validation steps are critical to making sure the system is operating correctly.
"You're replacing the microscope with a digital system. You want to be sure it is equivalent in accuracy to the glass slide and microscope," he explained.
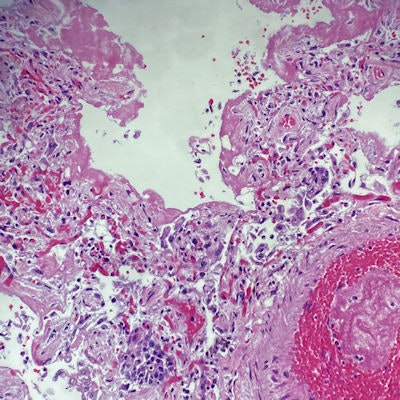
 Lung sections from a patient with SARS-CoV-2 show a prominent layer of eosinophilic material lining the alveolar membranes corresponding to the hyaline membrane, a hallmark of acute phase diffuse alveolar damage. Capillary congestion and microthromboemboli are also appreciated, supporting the diagnosis. Credit: Dr. Emma Henrie.
Lung sections from a patient with SARS-CoV-2 show a prominent layer of eosinophilic material lining the alveolar membranes corresponding to the hyaline membrane, a hallmark of acute phase diffuse alveolar damage. Capillary congestion and microthromboemboli are also appreciated, supporting the diagnosis. Credit: Dr. Emma Henrie.While there would be some costs associated with security and Health Insurance Portability and Accountability Act (HIPAA) compliance, these should not amount to a significant expense. As a way to hold down costs, the necessary software and hardware could even be leased, Glassy indicated.
"If you have the best person in your pathology group quarantined, then there would be an economic advantage to setting that person up to review cases remotely," he added.
Pixel depth
There might be monitor issues involving having enough pixel depth, as well concerns over having a fast-enough internet connection. Pathology images are big and require a lot of bandwith for remote reading. Recently, the U.S. Food and Drug Administration (FDA) granted waivers to allow the use of consumer monitors, instead of medical-grade monitors, for remote reading. Still, the entire system must undergo verification and validation, Glassy said.
"If you have a validated system at the hospital or main lab, then the cost of digitally working remotely can be quite low," he explained. "If not, then you have hardware and infrastructure expenses, as well as the need for validation and documentation."
Plan ahead
Although pathologists at Glassy's Affiliated Pathologists Medical Group are not yet working remotely, he said the group anticipates some might. So, the practice is putting in place policy procedures that would allow pathologists to read remotely, such as from their homes.
"We are thinking through this in a logical fashion and making sure all the needed documentation is in place, so the change could occur without a lot of disruption," he said.
But some pathologists may find the use of a computer and monitor disconcerting, compared with using a microscope and slides. There is an adjustment period for many pathologists, said Dr. Adam Booth, a resident physician in the pathology department at the University of Texas Medical Branch at Galveston.
"Some pathologists may be ready for the transition, but there will be a lag time for some others," Booth commented in an interview.
Budget concerns and secure data storage are also among the issues pathology groups may face, he added, echoing Glassy's sentiments.
Getting a COVID-19 push
Yet, Booth indicated that COVID-19 may push some of those issues aside and hasten transitions to telepathology and the remote workplace.
"It's important for pathologists to work at home if they are ill," he said. "They can review their cases at home and can order chemistries or any special stains they need. Then they can follow up on those cases and do remote sign-outs. This capability wouldn't delay a patient's diagnosis and would help guide a clinician's next steps."
Glassy and Booth both believe that the coronavirus will hasten acceptance of telepathology, not only for the remote reading of slides but also for teaching, continuing education, and training. Although those efforts may not bring in much revenue to a pathology practice, they would add to the professionalism and prestige of practices, they suggested.
Editor's note: This article is the first segment of a two-part series on telepathology. We explore ways telepathology is being put into practice in part 2.



















