More than 40 experts and 13 institutions are collaborating on a five-year, $41 million study funded by the National Institutes of Health (NIH) National Institute on Aging aimed at better understanding the biological pathways underlying Alzheimer’s disease.
The study, titled Centrally-linked Longitudinal Peripheral Biomarkers of AD (CLEAR-AD), may ultimately lead to more personalized patient care through the development of a blood test for multiple pathways implicated in the disease, thus enabling earlier and less-invasive diagnosis, the researchers said.
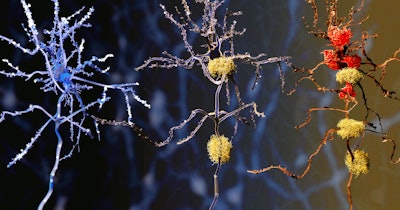
Most Alzheimer's disease diagnostic modalities focus on the hallmark amyloid plaques and tau tangles that build up in the brain. CLEAR-AD’s systems biology approach may help create an understanding of the factors that drive these pathological hallmarks in the first place. CLEAR-AD will include three closely related projects focusing on single-cell analysis of human brain tissue, longitudinal brain imaging, and blood-based biomarker research.
The first project will investigate brain tissue with single-cell and single-nucleus sequencing to understand the differences in gene expression across different types of brain cells from different brain regions. A database of molecular signatures connecting brain and blood will be produced for the research community to further analyze.
The second project will focus on longitudinal analysis of endophenotypes, measurable biological traits related to Alzheimer’s disease that can be studied with MRI and PET brain imaging, fluid biomarkers, and clinical tests including cognitive performance. Alzheimer’s disease hallmarks, including cognitive decline, loss of cortical thickness and hippocampal volume on MRI images, and accumulation of amyloid plaque and tau tangles on brain PET scans, will be related to molecular signatures from brain cells obtained in the first project. The findings may yield new insights into stage-specific disease evolution and indicate potential therapeutic targets.
The third project seeks to bridge current gaps in knowledge about disproportionately impacted communities of color, as past Alzheimer’s research predominately included data from white participants. The research will connect molecular signatures and endophenotypes from the first and second projects with patterns observed among African American and Hispanic participants, who are more likely to develop the disease but less likely to be diagnosed. CLEAR-AD will connect with local communities to uncover the differences in Alzheimer’s disease genes and endophenotypes, including imaging, cognition, and blood biomarkers, between people of color and their white counterparts.
The resulting data on all projects will be shared with the scientific community to accelerate collaborative progress. The researchers said they hope the large-scale data will advance precision medicine for patients through the development of a blood test for multiple pathways implicated in the disease.
“CLEAR-AD will allow us to connect what we see in peripheral blood and on imaging to what we see in brain tissue, which will be paramount in the opportunity to create the next generation of blood tests to diagnose individuals with Alzheimer’s disease even earlier, when the disease is most treatable,” Kwangsik Nho, PhD, the principal investigator and an associate professor at Indiana University School of Medicine, said in a statement.